Please select the key messages you would like to include in your personalized leaflet, by ticking the boxes
Quantity & Quality of weight loss


Abbreviation: MACE: Major Adverse Cardiovascular Event; PwO: People with Obesity
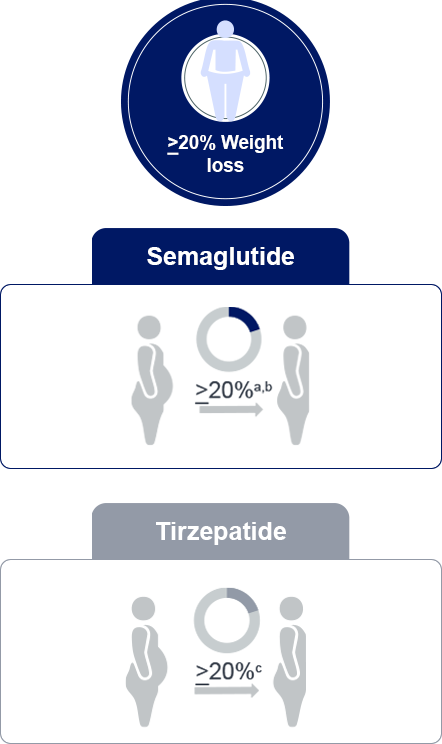
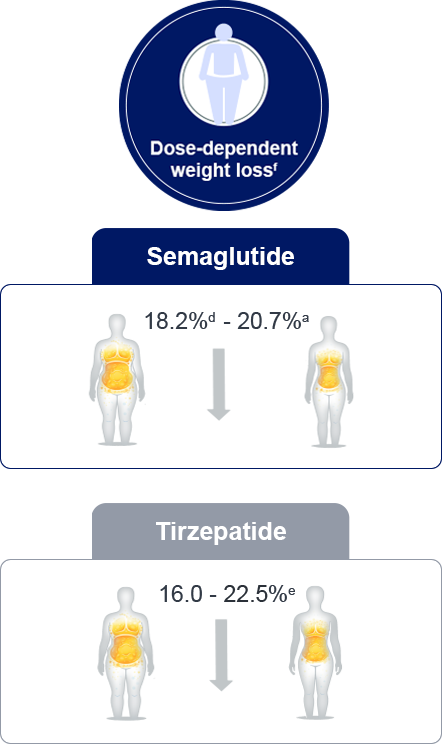
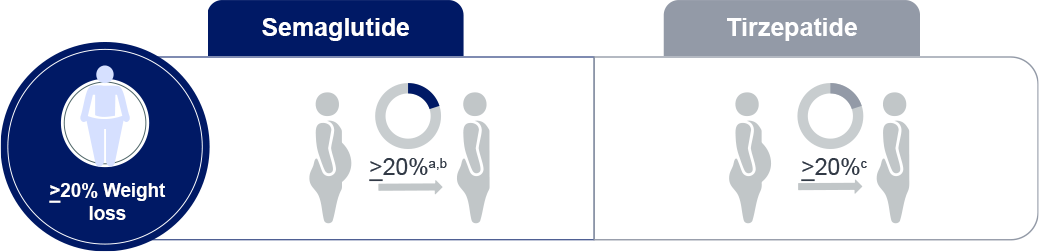
a: In STEP-UP trial, semaglutide 7.2 mg reduced weight by 20.7%
b: Across STEP trials, one-third of those on semaglutide 2.4 mg dose lost at least 20% of their
weight
c: SURMOUNT-1; Tirzepatide 10mg and 15mg
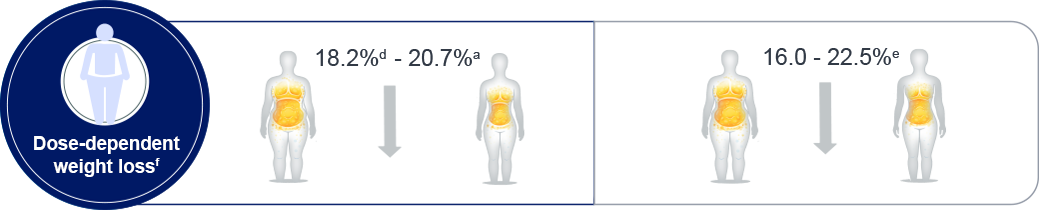
d: STEP 4 trial
e: SURMOUNT-1: Tirzepatide 5mg, 10 mg and 15 mg
f: Trial product estimand; Weight loss range is quoted by the minimum and maximum maintenance doses from
double-blind, placebo-controlled phase 3 trials
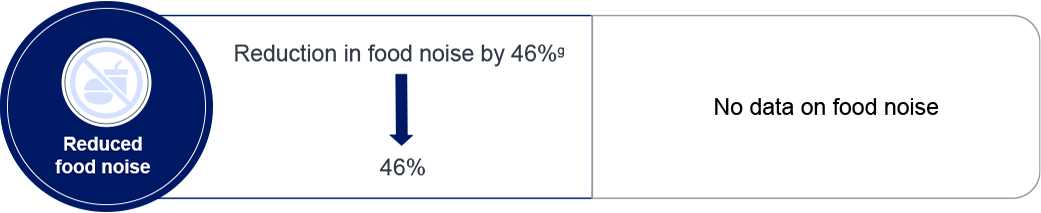
g: Arnaut T, et al. Presented at EASD 2025, Vienna, Austria, 15–19 September 2025
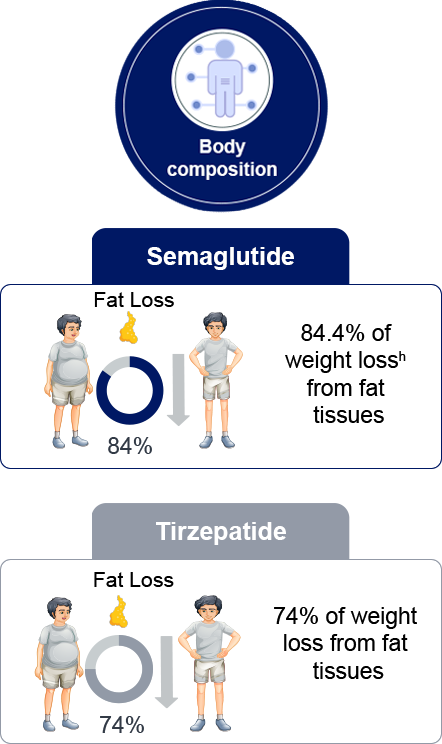
h: Hjelmesaeth J et al. Presented at the European Association for the Study of Diabetes (EASD) 61st
Annual Meeting. 15-19 September 2025; Vienna, Austria. Please note that Semaglutide 7.2 mg has been
approved by EMA but is not yet approved in Malaysia . This data is from a pooled population of
Semaglutide 2.4mg and 7.2mg from STEP UP trial.
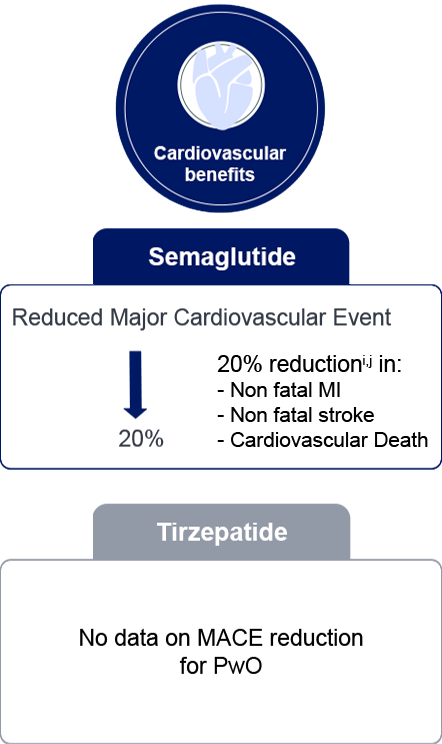
i: Semaglutide 2.4mg is currently not approved for prevention of cardiovascular disease in Malaysia.
Please refer to full prescribing information for more details.
j: Lincoff AM, et al. Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes. N Engl J Med.
2023;389:2221-32